Fast-Moving Trends in Drug Discovery and Development
Learn how new trends in drug discovery and alternatives to animal testing are accelerating drug development.
Featured Story
Learn how new trends in drug discovery and alternatives to animal testing are accelerating drug development.

3D Models in Drug Discovery Transform Advanced Therapeutics
3D models play an increasingly prominent role in drug discovery and cell therapy. Discover the latest 3D cell culture trends and how to address the challenges.
3D models play an...
3D models play an increasingly prominent...
3D models play an increasingly prominent role in drug discovery and cell...
3D models play an increasingly prominent role in drug discovery and cell therapy. Discover the latest 3D cell...
3D models play an increasingly prominent role in drug discovery and cell therapy. Discover the latest 3D cell culture trends and how to address...

Supporting a Maturing Modality
Advancing mesenchymal stem cell (MSC) therapies requires scalable manufacturing. Explore key challenges, quality considerations, and technologies enabling large-volume MSC production.
Advancing mesenchymal stem...
Advancing mesenchymal stem cell (MSC)...
Advancing mesenchymal stem cell (MSC) therapies requires scalable...
Advancing mesenchymal stem cell (MSC) therapies requires scalable manufacturing. Explore key challenges, quality...
Advancing mesenchymal stem cell (MSC) therapies requires scalable manufacturing. Explore key challenges, quality considerations, and...
High-Yield MSC Expansion at the Forefront of Cell Therapy Advancement
Discover how to achieve scalable, high-yield MSC expansion with Corning HYPERStack vessels and MSCulture Max media in closed, efficient workflows from bench to production.
Discover how to achieve...
Discover how to achieve scalable, high-yield...
Discover how to achieve scalable, high-yield MSC expansion with Corning...
Discover how to achieve scalable, high-yield MSC expansion with Corning HYPERStack vessels and MSCulture Max media...
Discover how to achieve scalable, high-yield MSC expansion with Corning HYPERStack vessels and MSCulture Max media in closed, efficient...

Scale Up Spheroid Production: FibroBiologics' Path to Clinical Scale
Corning's Elplasia technology is helping FibroBiologics produce 4.5 million uniform spheroids for the first spheroid-based therapeutic in a human clinical trial.
Corning's Elplasia technology...
Corning's Elplasia technology is helping...
Corning's Elplasia technology is helping FibroBiologics produce 4.5...
Corning's Elplasia technology is helping FibroBiologics produce 4.5 million uniform spheroids for the first...
Corning's Elplasia technology is helping FibroBiologics produce 4.5 million uniform spheroids for the first spheroid-based therapeutic in a...

Scaling MSC Therapies: Overcoming Risks and Barriers
Scaling MSC therapies is simpler and less risky with the right tools, such as stacked modular cell culture systems and closed-system solutions.
Scaling MSC therapies is...
Scaling MSC therapies is simpler and less...
Scaling MSC therapies is simpler and less risky with the right tools, such...
Scaling MSC therapies is simpler and less risky with the right tools, such as stacked modular cell culture systems...
Scaling MSC therapies is simpler and less risky with the right tools, such as stacked modular cell culture systems and closed-system...
Comparing MSC Isolation Techniques
Comparing mesenchymal stem cell (MSC) isolation techniques. Learn the specific advantages and limitations of enzymatic and explant culture methods. Comparing MSC isolation techniques.
Comparing mesenchymal stem...
Comparing mesenchymal stem cell (MSC)...
Comparing mesenchymal stem cell (MSC) isolation techniques. Learn the...
Comparing mesenchymal stem cell (MSC) isolation techniques. Learn the specific advantages and limitations of...
Comparing mesenchymal stem cell (MSC) isolation techniques. Learn the specific advantages and limitations of enzymatic and explant culture...
Fast-Moving Trends in Drug Discovery and Development
Learn how new trends in drug discovery and alternatives to animal testing are accelerating drug development.
Learn how new trends in drug...
Learn how new trends in drug discovery and...
Learn how new trends in drug discovery and alternatives to animal testing...
Learn how new trends in drug discovery and alternatives to animal testing are accelerating drug development.
Learn how new trends in drug discovery and alternatives to animal testing are accelerating drug development.
What are MSCs and How are They Used?
Learn more about mesenchymal stem cells (MSCs), where they come from, and their traditional and evolving clinical applications and therapies.
Learn more about mesenchymal...
Learn more about mesenchymal stem cells...
Learn more about mesenchymal stem cells (MSCs), where they come from, and...
Learn more about mesenchymal stem cells (MSCs), where they come from, and their traditional and evolving clinical...
Learn more about mesenchymal stem cells (MSCs), where they come from, and their traditional and evolving clinical applications and therapies.

Producing MSC-derived cell therapies: workflows, technologies, and case studies
In this expert roundtable, four highly experienced industry professionals discuss key considerations in mesenchymal stem (or stromal) cell (MSC) manufacturing, including reagent selection, dosing, delivery methods, and final product formulation.
In this expert roundtable,...
In this expert roundtable, four highly...
In this expert roundtable, four highly experienced industry professionals...
In this expert roundtable, four highly experienced industry professionals discuss key considerations in...
In this expert roundtable, four highly experienced industry professionals discuss key considerations in mesenchymal stem (or stromal) cell (MSC)...
Methods for MSC Concentration and Buffer Exchange
Learn more about mesenchymal stem/stromal cell concentration and buffers exchange techniques to ensure an informed choice balancing method performance and cost.
Learn more about mesenchymal...
Learn more about mesenchymal stem/stromal...
Learn more about mesenchymal stem/stromal cell concentration and buffers...
Learn more about mesenchymal stem/stromal cell concentration and buffers exchange techniques to ensure an informed...
Learn more about mesenchymal stem/stromal cell concentration and buffers exchange techniques to ensure an informed choice balancing method...

Corning 3D Cell Culture Summit 2024
At the Corning 3D Cell Culture Summit 2024, scientists presented the latest 3D culture research and tools and discussed best practices for 3D culture.
At the Corning 3D Cell...
At the Corning 3D Cell Culture Summit 2024,...
At the Corning 3D Cell Culture Summit 2024, scientists presented the...
At the Corning 3D Cell Culture Summit 2024, scientists presented the latest 3D culture research and tools and...
At the Corning 3D Cell Culture Summit 2024, scientists presented the latest 3D culture research and tools and discussed best practices for 3D...

Integrating Next-Generation Sequencing into Drug Discovery
Next-generation sequencing (NGS) has increasing roles in drug discovery and precision medicine and is benefiting from innovations in data analysis.
Next-generation sequencing...
Next-generation sequencing (NGS) has...
Next-generation sequencing (NGS) has increasing roles in drug discovery...
Next-generation sequencing (NGS) has increasing roles in drug discovery and precision medicine and is benefiting...
Next-generation sequencing (NGS) has increasing roles in drug discovery and precision medicine and is benefiting from innovations in data...

7 Fast Facts About PYREX Lab Glass
Explore the facts about PYREX lab glass, like chemical resistance and standardization, that make it essential lab equipment.
Explore the facts about PYREX...
Explore the facts about PYREX lab glass,...
Explore the facts about PYREX lab glass, like chemical resistance and...
Explore the facts about PYREX lab glass, like chemical resistance and standardization, that make it essential lab...
Explore the facts about PYREX lab glass, like chemical resistance and standardization, that make it essential lab equipment.

Patient-Derived Organoids Are Pioneering Personalized Treatment Strategies
Patient-derived organoids are advancing precision medicine by helping predict patients' responses to therapies.
Patient-derived organoids are...
Patient-derived organoids are advancing...
Patient-derived organoids are advancing precision medicine by helping...
Patient-derived organoids are advancing precision medicine by helping predict patients' responses to therapies.
Patient-derived organoids are advancing precision medicine by helping predict patients' responses to therapies.
Coatings for Bioreactor Scale-up vs. Other Stem Cell Production Systems
Options for large-volume stem cell growth include microcarrier or bioreactor scale-up and stacked vessel scale-out. The choice of vessel coating is key.
Options for large-volume stem...
Options for large-volume stem cell growth...
Options for large-volume stem cell growth include microcarrier or...
Options for large-volume stem cell growth include microcarrier or bioreactor scale-up and stacked vessel...
Options for large-volume stem cell growth include microcarrier or bioreactor scale-up and stacked vessel scale-out. The choice of vessel coating...
The Benefits of Coatings for Bioproduction: Stem Cells and Coatings
Finding the right coating is critical for stem cell production. Benefits of coatings for bio production include attachment, signaling, and cell fate.
Finding the right coating is...
Finding the right coating is critical for...
Finding the right coating is critical for stem cell production. Benefits...
Finding the right coating is critical for stem cell production. Benefits of coatings for bio production include...
Finding the right coating is critical for stem cell production. Benefits of coatings for bio production include attachment, signaling, and cell...

Overcoming Bioprocessing Bottlenecks By Combining Adherent And Suspension Systems
Waisman Biomanufacturing has released the results of an internal study where they demonstrate comparability for a new manufacturing platform combining bioreactors and microcarriers using MSCs.
Waisman Biomanufacturing has...
Waisman Biomanufacturing has released the...
Waisman Biomanufacturing has released the results of an internal study...
Waisman Biomanufacturing has released the results of an internal study where they demonstrate comparability for a...
Waisman Biomanufacturing has released the results of an internal study where they demonstrate comparability for a new manufacturing platform...
Dynamic Cell Cultures: Organoids and Spheroids in Suspension
Advances in dynamic cell cultures now allow large numbers of organoids and spheroids to be generated in suspension. Learn more.
Advances in dynamic cell...
Advances in dynamic cell cultures now allow...
Advances in dynamic cell cultures now allow large numbers of organoids and...
Advances in dynamic cell cultures now allow large numbers of organoids and spheroids to be generated in...
Advances in dynamic cell cultures now allow large numbers of organoids and spheroids to be generated in suspension. Learn more.

Fast-Moving Trends in Drug Discovery
Drug discovery is changing with the rapid growth of AI, the availability of more realistic drug screening environments, and the rise of precision medicine.
Drug discovery is changing...
Drug discovery is changing with the rapid...
Drug discovery is changing with the rapid growth of AI, the availability...
Drug discovery is changing with the rapid growth of AI, the availability of more realistic drug screening...
Drug discovery is changing with the rapid growth of AI, the availability of more realistic drug screening environments, and the rise of...

In-House vs. CDMO Cell Therapy Development: Which Is Right for Your Lab?
Cell therapy labs need to weigh the benefits and drawbacks of in-house vs. CDMO cell therapy development and manufacturing.
Cell therapy labs need to...
Cell therapy labs need to weigh the benefits...
Cell therapy labs need to weigh the benefits and drawbacks of in-house vs....
Cell therapy labs need to weigh the benefits and drawbacks of in-house vs. CDMO cell therapy development and...
Cell therapy labs need to weigh the benefits and drawbacks of in-house vs. CDMO cell therapy development and manufacturing.

What to Know About CRISPR Screens and Bulk Spheroid Production
Cancer spheroids are an up-and-coming group of models with increasing uses in research, including CRISPR screens. Here's what to know.
Cancer spheroids are an...
Cancer spheroids are an up-and-coming group...
Cancer spheroids are an up-and-coming group of models with increasing uses...
Cancer spheroids are an up-and-coming group of models with increasing uses in research, including CRISPR screens....
Cancer spheroids are an up-and-coming group of models with increasing uses in research, including CRISPR screens. Here's what to know.
A Guide to Synthetic Hydrogels for 3D Cell Culture
Hydrogels provide a customizable support matrix for 3D cell culture that allows cells to behave naturally. Here's your guide to synthetic hydrogels.
Hydrogels provide a...
Hydrogels provide a customizable support...
Hydrogels provide a customizable support matrix for 3D cell culture that...
Hydrogels provide a customizable support matrix for 3D cell culture that allows cells to behave naturally. Here's...
Hydrogels provide a customizable support matrix for 3D cell culture that allows cells to behave naturally. Here's your guide to synthetic...

How CDMOs Have Become Emerging Technology Influencers
Learn how CDMOs fill the gap between science and commercial sectors to maximize the manufacturing process of cell-and-gene therapeutics.
Learn how CDMOs fill the gap...
Learn how CDMOs fill the gap between science...
Learn how CDMOs fill the gap between science and commercial sectors to...
Learn how CDMOs fill the gap between science and commercial sectors to maximize the manufacturing process of...
Learn how CDMOs fill the gap between science and commercial sectors to maximize the manufacturing process of cell-and-gene therapeutics.
Precision Medicine Advancements: The Role of 3D Cell Culture
Learn how technological advancements, like 3D cell culture, have opened the doors to faster, more reliable drug development.
Learn how technological...
Learn how technological advancements, like...
Learn how technological advancements, like 3D cell culture, have opened...
Learn how technological advancements, like 3D cell culture, have opened the doors to faster, more reliable drug...
Learn how technological advancements, like 3D cell culture, have opened the doors to faster, more reliable drug development.
Cut the Supply Chain Plastic Footprint With Innovation and Intensification
Innovation and intensifying production can help reduce the plastic footprint of laboratory practices for more sustainability, efficiency, and cost savings.
Innovation and intensifying...
Innovation and intensifying production can...
Innovation and intensifying production can help reduce the plastic...
Innovation and intensifying production can help reduce the plastic footprint of laboratory practices for more...
Innovation and intensifying production can help reduce the plastic footprint of laboratory practices for more sustainability, efficiency, and...

Digital Transformation With New Laboratory Technology
Digital transformation with new laboratory technology helps reduce errors, speed up lab functions and optimize data handling.
Digital transformation with...
Digital transformation with new laboratory...
Digital transformation with new laboratory technology helps reduce errors,...
Digital transformation with new laboratory technology helps reduce errors, speed up lab functions and optimize...
Digital transformation with new laboratory technology helps reduce errors, speed up lab functions and optimize data handling.

Corning’s Axygen® Microtube Shaker Marries Uniformity and Compaction
The Axygen Microtube Shaker is a benchtop shaking incubator allowing uniform heating, cooling, and mixing of precious life science.
The Axygen Microtube Shaker...
The Axygen Microtube Shaker is a benchtop...
The Axygen Microtube Shaker is a benchtop shaking incubator allowing...
The Axygen Microtube Shaker is a benchtop shaking incubator allowing uniform heating, cooling, and mixing of...
The Axygen Microtube Shaker is a benchtop shaking incubator allowing uniform heating, cooling, and mixing of precious life science.
Gene Therapies: From Research To Reality
Due to the complexity of cell and gene therapy products and manufacturing processes, it is crucial to conduct due diligence before committing to a partner.
Due to the complexity of cell...
Due to the complexity of cell and gene...
Due to the complexity of cell and gene therapy products and manufacturing...
Due to the complexity of cell and gene therapy products and manufacturing processes, it is crucial to conduct due...
Due to the complexity of cell and gene therapy products and manufacturing processes, it is crucial to conduct due diligence before committing to...

How Collaborative R&D Delivered Revolutionary Adherent Culture Scale-Up Technology
The Corning® Ascent® FBR System can ease or eliminate the difficult choice between adherent and suspension cell culture platforms.
The Corning® Ascent® FBR...
The Corning® Ascent® FBR System can ease or...
The Corning® Ascent® FBR System can ease or eliminate the difficult choice...
The Corning® Ascent® FBR System can ease or eliminate the difficult choice between adherent and suspension cell...
The Corning® Ascent® FBR System can ease or eliminate the difficult choice between adherent and suspension cell culture platforms.

Scale-Up Technology for AAV Production
Corning customer Dyno Therapeutics is leveraging Corning’s Ascent Fixed Bed Bioreactor system to efficiently scale-up AAV production.
Corning customer Dyno...
Corning customer Dyno Therapeutics is...
Corning customer Dyno Therapeutics is leveraging Corning’s Ascent Fixed...
Corning customer Dyno Therapeutics is leveraging Corning’s Ascent Fixed Bed Bioreactor system to efficiently...
Corning customer Dyno Therapeutics is leveraging Corning’s Ascent Fixed Bed Bioreactor system to efficiently scale-up AAV production.
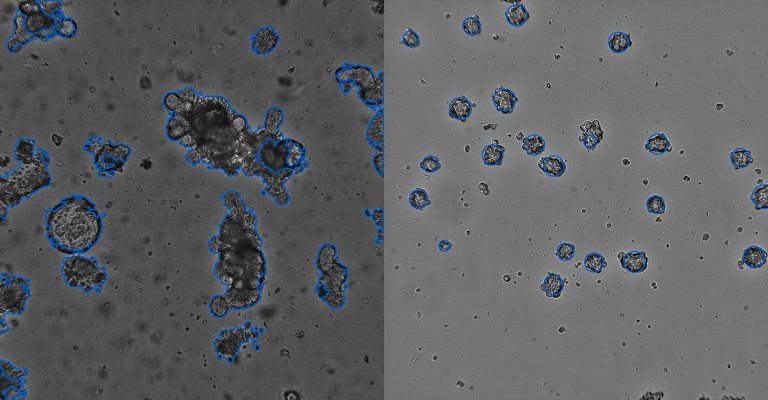
New Corning Solution Streamlines 3D Cell Culture Analysis
Organoid Counting Software for the Corning Cell Counter meets the unique needs of researchers working with more complex and predictive 3D cell cultures.
Organoid Counting Software...
Organoid Counting Software for the Corning...
Organoid Counting Software for the Corning Cell Counter meets the unique...
Organoid Counting Software for the Corning Cell Counter meets the unique needs of researchers working with more...
Organoid Counting Software for the Corning Cell Counter meets the unique needs of researchers working with more complex and predictive 3D cell...
3D Cell Culture Promises Tissue Model Success for Drug Screening
Drug screening is an expensive process that requires many different steps for success. Tissue model technology is helping to reduce the costs.
Drug screening is an...
Drug screening is an expensive process that...
Drug screening is an expensive process that requires many different steps...
Drug screening is an expensive process that requires many different steps for success. Tissue model technology is...
Drug screening is an expensive process that requires many different steps for success. Tissue model technology is helping to reduce the costs.

Viral Vector Manufacturing and Cell Harvesting from a Single Bioreactor?
Viral Vector Manufacturing and Cell Harvesting from a Single Bioreactor? Increased cell productivity translates to more product from fewer culture cycles.
Viral Vector Manufacturing...
Viral Vector Manufacturing and Cell...
Viral Vector Manufacturing and Cell Harvesting from a Single Bioreactor?...
Viral Vector Manufacturing and Cell Harvesting from a Single Bioreactor? Increased cell productivity translates to...
Viral Vector Manufacturing and Cell Harvesting from a Single Bioreactor? Increased cell productivity translates to more product from fewer...
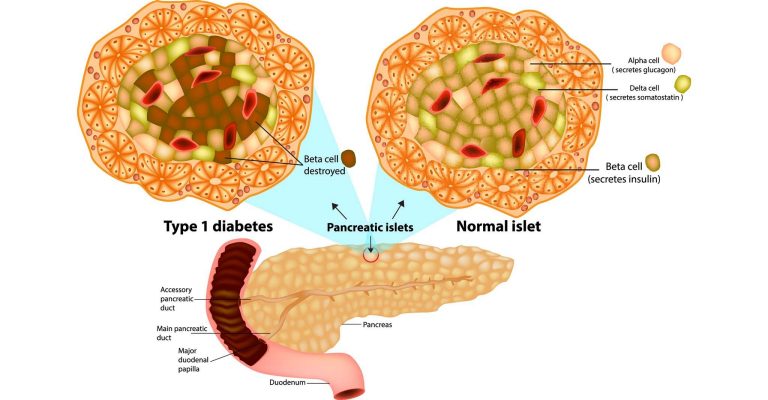
A Novel Approach to Type 1 Diabetes Treatment
A novel viral vector approach is giving new hope for a breakthrough diabetes treatment. Efficient scale-up technology could help make this a reality.
A novel viral vector approach...
A novel viral vector approach is giving new...
A novel viral vector approach is giving new hope for a breakthrough...
A novel viral vector approach is giving new hope for a breakthrough diabetes treatment. Efficient scale-up...
A novel viral vector approach is giving new hope for a breakthrough diabetes treatment. Efficient scale-up technology could help make this a...

Identifying Tumor-Specific Cancer Treatments Using Patient-Derived Tumor Organoids
Learn how pairing genomics with drug screening of high-fidelity patient-derived organoids may identify effective therapeutic strategies for cancer patients.
Learn how pairing genomics...
Learn how pairing genomics with drug...
Learn how pairing genomics with drug screening of high-fidelity...
Learn how pairing genomics with drug screening of high-fidelity patient-derived organoids may identify effective...
Learn how pairing genomics with drug screening of high-fidelity patient-derived organoids may identify effective therapeutic strategies for...
Cancer Treatment and Other Therapies Impacted by Stem Cell Source
Stem cell source may impact cancer treatment and MSC therapy success. Researchers must consider cell characteristics and expansion potential when sourcing.
Stem cell source may impact...
Stem cell source may impact cancer treatment...
Stem cell source may impact cancer treatment and MSC therapy success....
Stem cell source may impact cancer treatment and MSC therapy success. Researchers must consider cell...
Stem cell source may impact cancer treatment and MSC therapy success. Researchers must consider cell characteristics and expansion potential...
Cryopreservation in Stem Cell Therapy
Stem cell cryopreservation for cell-based therapies allows scientists to pause cell culture for flexibility during characterization and scaling up.
Stem cell cryopreservation...
Stem cell cryopreservation for cell-based...
Stem cell cryopreservation for cell-based therapies allows scientists to...
Stem cell cryopreservation for cell-based therapies allows scientists to pause cell culture for flexibility during...
Stem cell cryopreservation for cell-based therapies allows scientists to pause cell culture for flexibility during characterization and scaling...

Why Are MSC-EVs Important and How Can You Scale-Up?
Mesenchymal stem cells extracellular vesicles (MSC-EVs) are emerging as valuable acellular therapeutics that avoid MSC safety issues.
Mesenchymal stem cells...
Mesenchymal stem cells extracellular...
Mesenchymal stem cells extracellular vesicles (MSC-EVs) are emerging as...
Mesenchymal stem cells extracellular vesicles (MSC-EVs) are emerging as valuable acellular therapeutics that avoid...
Mesenchymal stem cells extracellular vesicles (MSC-EVs) are emerging as valuable acellular therapeutics that avoid MSC safety issues.
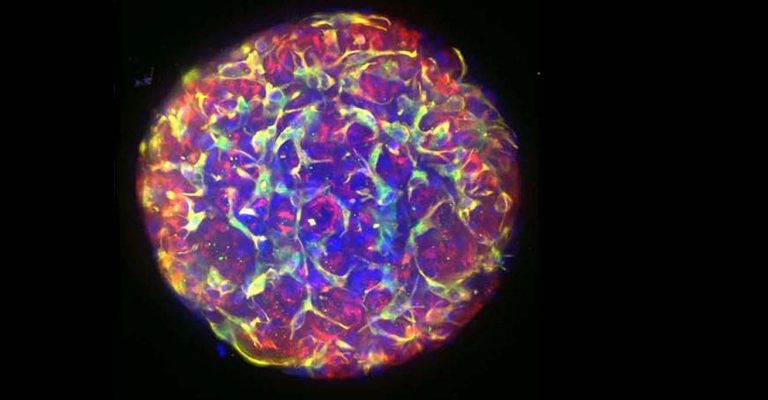
Best Practices for 3D Cell Imaging
Best practices in cell imaging of 3D cultures, like spheroids and organoids, include tissue preparation to aid light penetration and avoid sampling bias.
Best practices in cell...
Best practices in cell imaging of 3D...
Best practices in cell imaging of 3D cultures, like spheroids and...
Best practices in cell imaging of 3D cultures, like spheroids and organoids, include tissue preparation to aid...
Best practices in cell imaging of 3D cultures, like spheroids and organoids, include tissue preparation to aid light penetration and avoid...

Bringing 3D Organoid Models Into a High-Throughput Environment
Three-dimensional (3D) cell culture models, including organoids, are expected to increase the quality of compounds in the drug development pipeline, as they are poised to become the most faithful representation of the in vivo environment that can be studied in vitro.
Three-dimensional (3D) cell...
Three-dimensional (3D) cell culture models,...
Three-dimensional (3D) cell culture models, including organoids, are...
Three-dimensional (3D) cell culture models, including organoids, are expected to increase the quality of compounds...
Three-dimensional (3D) cell culture models, including organoids, are expected to increase the quality of compounds in the drug development...

Optimizing Vaccine Production Through Customization
Single-use technologies and modular production facilities may be the future of the most efficient and cost-effective vaccine development.
Single-use technologies and...
Single-use technologies and modular...
Single-use technologies and modular production facilities may be the...
Single-use technologies and modular production facilities may be the future of the most efficient and...
Single-use technologies and modular production facilities may be the future of the most efficient and cost-effective vaccine development.

Transfection Best Practices for AAV Gene Therapy Programs
Transfection strategies for gene therapy programs can have implications for cost and scale. There are several approaches for scientists to consider.
Transfection strategies for...
Transfection strategies for gene therapy...
Transfection strategies for gene therapy programs can have implications...
Transfection strategies for gene therapy programs can have implications for cost and scale. There are several...
Transfection strategies for gene therapy programs can have implications for cost and scale. There are several approaches for scientists to...
The Growing Role of MSCs in Regenerative Medicine
Despite the promise MSCs have in regenerative medicine, there are challenges of scale to overcome. Tools, technologies, and standardization can help.
Despite the promise MSCs have...
Despite the promise MSCs have in...
Despite the promise MSCs have in regenerative medicine, there are...
Despite the promise MSCs have in regenerative medicine, there are challenges of scale to overcome. Tools,...
Despite the promise MSCs have in regenerative medicine, there are challenges of scale to overcome. Tools, technologies, and standardization can...
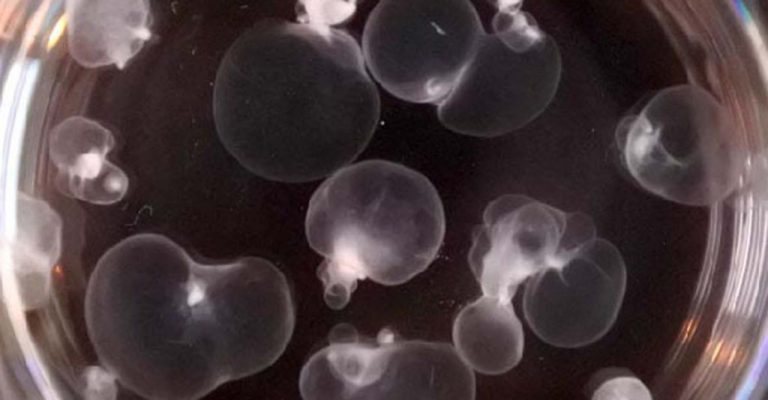
Organoids and Organ Regeneration: Cracking the Code
Organoids have had a profound effect on our understanding of human biology—particularly in the field of regenerative medicine.
Organoids have had a profound...
Organoids have had a profound effect on our...
Organoids have had a profound effect on our understanding of human...
Organoids have had a profound effect on our understanding of human biology—particularly in the field of...
Organoids have had a profound effect on our understanding of human biology—particularly in the field of regenerative medicine.

3D Cell Culture: From Academia to Big Pharma
Once on the academic fringes, 3D cell culture techniques have become popular in big pharma. But a few barriers still stand in the way of total adoption.
Once on the academic fringes,...
Once on the academic fringes, 3D cell...
Once on the academic fringes, 3D cell culture techniques have become...
Once on the academic fringes, 3D cell culture techniques have become popular in big pharma. But a few barriers...
Once on the academic fringes, 3D cell culture techniques have become popular in big pharma. But a few barriers still stand in the way of total...
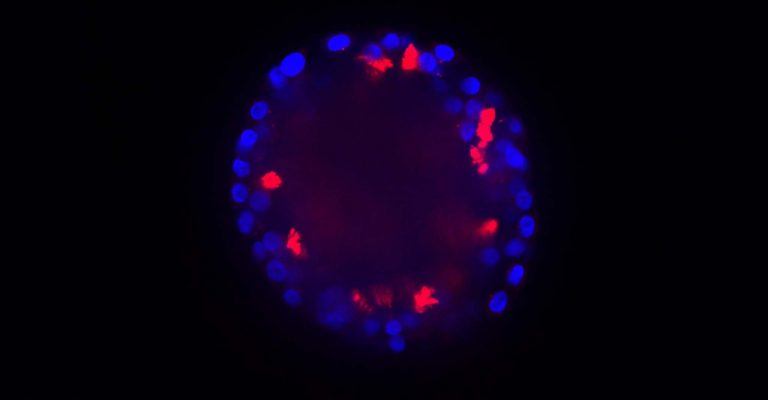
Disruptive Technologies in Life Sciences: Personalized Medicines
Corning Life Sciences is committed to enabling the accelerated development and commercialization of precision medicines.
Corning Life Sciences is...
Corning Life Sciences is committed to...
Corning Life Sciences is committed to enabling the accelerated development...
Corning Life Sciences is committed to enabling the accelerated development and commercialization of precision...
Corning Life Sciences is committed to enabling the accelerated development and commercialization of precision medicines.
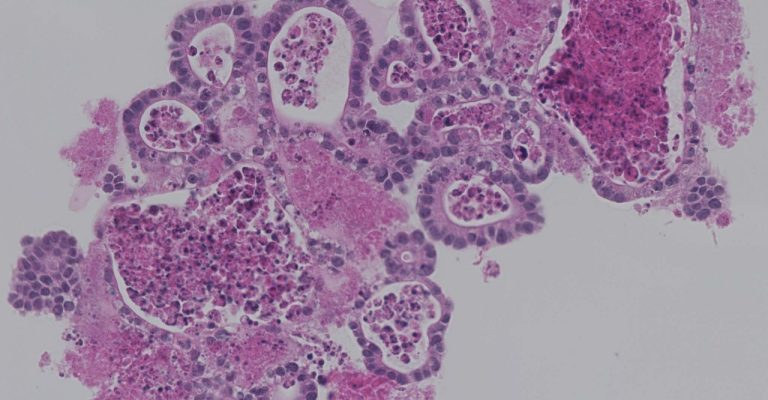
Spheroids, Organoids Replacing Standard Cultures for Cell-Based Assays
As 3D culture has grown in popularity, the terms “spheroid” and “organoid” have been used interchangeably; however, the differences between these culture types are significant.
As 3D culture has grown in...
As 3D culture has grown in popularity, the...
As 3D culture has grown in popularity, the terms “spheroid” and “organoid”...
As 3D culture has grown in popularity, the terms “spheroid” and “organoid” have been used interchangeably;...
As 3D culture has grown in popularity, the terms “spheroid” and “organoid” have been used interchangeably; however, the differences between...
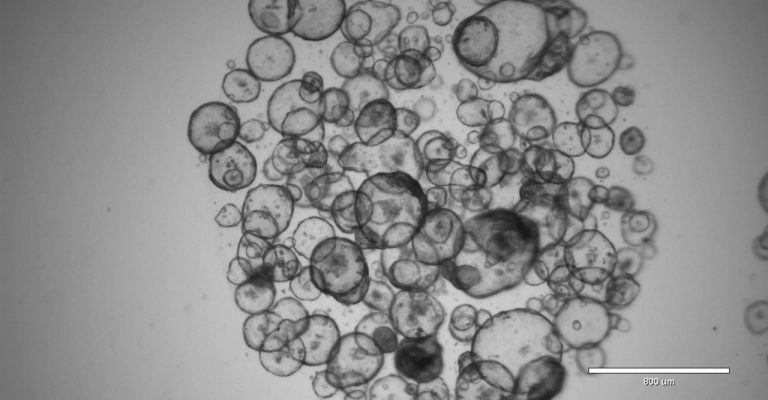
A 3D Cell Culture Primer: Freezing, Culturing, and Measuring Organoids
Engaging in 3D cell culture projects is thrilling but complex. Help your organoids thrive with these easy-to-follow best practices.
Engaging in 3D cell culture...
Engaging in 3D cell culture projects is...
Engaging in 3D cell culture projects is thrilling but complex. Help your...
Engaging in 3D cell culture projects is thrilling but complex. Help your organoids thrive with these...
Engaging in 3D cell culture projects is thrilling but complex. Help your organoids thrive with these easy-to-follow best practices.

Why We Should Be Interested in Human Tissue Chips
Dr. Kacey Ronaldson-Bouchard explains why adapting cell-based assays from suspension cells to 2D culture, 3D culture, and beyond to ‘patient in a dish’ systems is not an exercise undertaken lightly. However, the effort pays off on many levels.
Dr. Kacey Ronaldson-Bouchard...
Dr. Kacey Ronaldson-Bouchard explains why...
Dr. Kacey Ronaldson-Bouchard explains why adapting cell-based assays from...
Dr. Kacey Ronaldson-Bouchard explains why adapting cell-based assays from suspension cells to 2D culture, 3D...
Dr. Kacey Ronaldson-Bouchard explains why adapting cell-based assays from suspension cells to 2D culture, 3D culture, and beyond to ‘patient in...
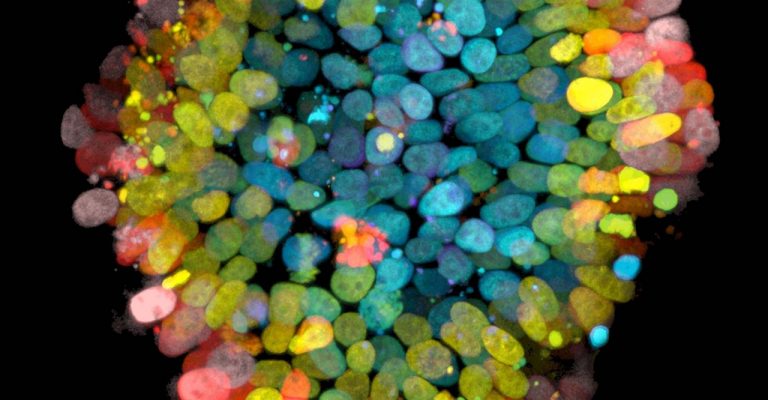
Using Organoids in the Study of Infectious Diseases
Organoid cell culture has transformed cell-based assays in drug discovery and basic biology by conferring physiologic relevance to in vitro cell-based biological models.
Organoid cell culture has...
Organoid cell culture has transformed...
Organoid cell culture has transformed cell-based assays in drug discovery...
Organoid cell culture has transformed cell-based assays in drug discovery and basic biology by conferring...
Organoid cell culture has transformed cell-based assays in drug discovery and basic biology by conferring physiologic relevance to in vitro...

Harvard Researcher Tackles the Big Question in Cancer Research: Drug Resistance
In this exclusive interview, we speak with Dr. Fang Xie, research fellow at Harvard Medical School’s Beth Israel Deaconess Medical Center, to find out more about his work investigating prostate cancer therapeutics, notably chemotherapy.
In this exclusive interview,...
In this exclusive interview, we speak with...
In this exclusive interview, we speak with Dr. Fang Xie, research fellow...
In this exclusive interview, we speak with Dr. Fang Xie, research fellow at Harvard Medical School’s Beth Israel...
In this exclusive interview, we speak with Dr. Fang Xie, research fellow at Harvard Medical School’s Beth Israel Deaconess Medical Center, to...
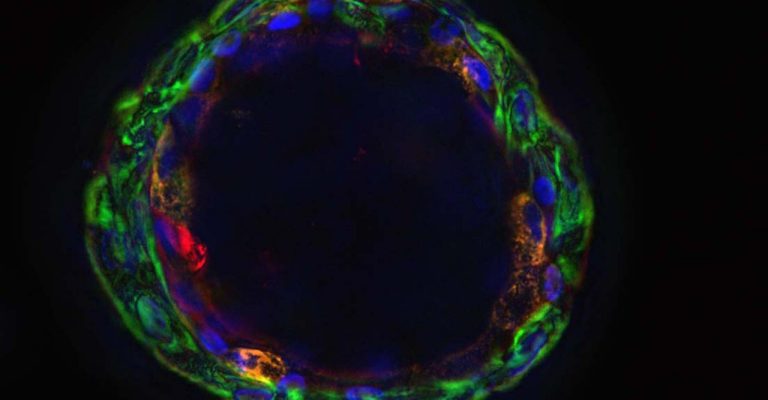
Ramping Up Drug, Vaccine and Infectivity Testing With Airway Organoids
Lung and airway organoids are of interest for both drug and vaccine development and are valuable tools for studying infectivity in human respiratory diseases, particularly for challenging viral diseases like COVID-19.
Lung and airway organoids are...
Lung and airway organoids are of interest...
Lung and airway organoids are of interest for both drug and vaccine...
Lung and airway organoids are of interest for both drug and vaccine development and are valuable tools for...
Lung and airway organoids are of interest for both drug and vaccine development and are valuable tools for studying infectivity in human...

Why HUB Organoids are Key to Advancing Personalized Cancer Therapies
Hubrecht Organoid Technology (HUB) details how patient-derived organoids are becoming promising models for personalized treatment using 3D cell culture technologies from Corning and Yamaha Corporation.
Hubrecht Organoid Technology...
Hubrecht Organoid Technology (HUB) details...
Hubrecht Organoid Technology (HUB) details how patient-derived organoids...
Hubrecht Organoid Technology (HUB) details how patient-derived organoids are becoming promising models for...
Hubrecht Organoid Technology (HUB) details how patient-derived organoids are becoming promising models for personalized treatment using 3D cell...

3D Cell Culture Could Drive the Fight Against Infectious Diseases
3D cell culture models are one of the technologies leading the fight against infectious diseases, which include the novel coronavirus, COVID-19.
3D cell culture models are...
3D cell culture models are one of the...
3D cell culture models are one of the technologies leading the fight...
3D cell culture models are one of the technologies leading the fight against infectious diseases, which include...
3D cell culture models are one of the technologies leading the fight against infectious diseases, which include the novel coronavirus,...

Harnessing Next-Generation Cell Culture Methods in the Fight Against Cancer
Learn how an institute in the US is developing the latest patient-derived 3D cancer cell cultures for use in precision medicine.
Learn how an institute in the...
Learn how an institute in the US is...
Learn how an institute in the US is developing the latest patient-derived...
Learn how an institute in the US is developing the latest patient-derived 3D cancer cell cultures for use in...
Learn how an institute in the US is developing the latest patient-derived 3D cancer cell cultures for use in precision medicine.
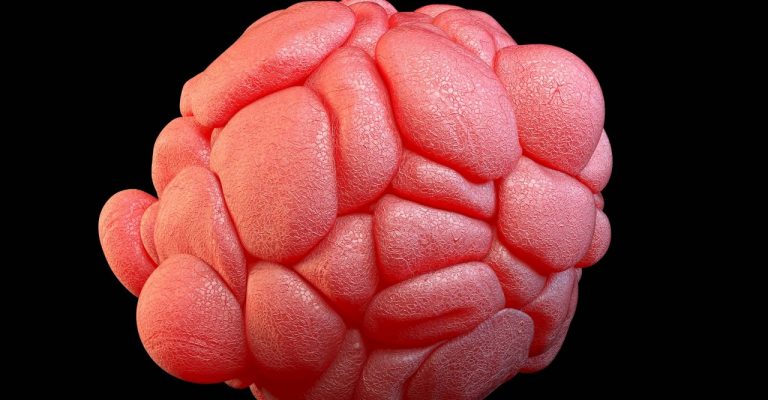
Cellular Scaffolding and Modern Tissue Engineering
Explore the differences in process when using a scaffold-based versus scaffold-free platform in tissue engineering. Learn about the emergence of scaffold-free platforms in the tissue engineering space, with success in musculoskeletal cartilages, for example.
Explore the differences in...
Explore the differences in process when...
Explore the differences in process when using a scaffold-based versus...
Explore the differences in process when using a scaffold-based versus scaffold-free platform in tissue...
Explore the differences in process when using a scaffold-based versus scaffold-free platform in tissue engineering. Learn about the emergence of...
The Future of Joint Replacement Is Tissue Engineering
Tissue engineering offers promise for the future of joint replacement. Learn about the role of 3D cells in tissue engineering and the 3D systems being researched to bring us closer to regenerating joints and avoiding surgery.
Tissue engineering offers...
Tissue engineering offers promise for the...
Tissue engineering offers promise for the future of joint replacement....
Tissue engineering offers promise for the future of joint replacement. Learn about the role of 3D cells in tissue...
Tissue engineering offers promise for the future of joint replacement. Learn about the role of 3D cells in tissue engineering and the 3D systems...

The 3D Cell Culture Market Is Set for Rapid, Sustained Growth
The 3D cell culture market is projected to grow significantly. Learn about market segmentation and dynamics, where the 3D cell culture market is set to grow, how it's estimated to happen, and in particular, the future of 3D cell culture with cell therapy/regenerative and personalized medicine.
The 3D cell culture market is...
The 3D cell culture market is projected to...
The 3D cell culture market is projected to grow significantly. Learn about...
The 3D cell culture market is projected to grow significantly. Learn about market segmentation and dynamics, where...
The 3D cell culture market is projected to grow significantly. Learn about market segmentation and dynamics, where the 3D cell culture market is...
Prostate Cancer Researchers Use Organoids to Identify Critical Risk Factors
Prostate cancer researchers now have a powerful new tool available: prostate organoids that can accurately mimic in vivo conditions. Learn what prostate organoids are, their development and how they test and treat prostate cancer.
Prostate cancer researchers...
Prostate cancer researchers now have a...
Prostate cancer researchers now have a powerful new tool available:...
Prostate cancer researchers now have a powerful new tool available: prostate organoids that can accurately mimic...
Prostate cancer researchers now have a powerful new tool available: prostate organoids that can accurately mimic in vivo conditions....
CAR-T Cells, Immunotherapy, and 3D Models Are Paving the Way in Cancer Research
This article explores the role of immunotherapy in cancer treatment: What are CAR-T cells? How are in vivo-like environments and CAR-T cells innovating cancer therapy? and What does the future hold for CAR-T cell immunotherapy in the 3D landscape?
This article explores the...
This article explores the role of...
This article explores the role of immunotherapy in cancer treatment: What...
This article explores the role of immunotherapy in cancer treatment: What are CAR-T cells? How are in...
This article explores the role of immunotherapy in cancer treatment: What are CAR-T cells? How are in vivo-like environments and CAR-T...
3D Cell Culture Applications and Bioprinting Will Change How We Research Cancers
With 3D cell culture applications and bioprinting, cancer research is changing. Learn more about the increasing relevance of 3D bioprinting, how it can be tailored to specific types of tissues, cells and cancer, and how 3D models provide summarization of different tumor biology aspects.
With 3D cell culture...
With 3D cell culture applications and...
With 3D cell culture applications and bioprinting, cancer research is...
With 3D cell culture applications and bioprinting, cancer research is changing. Learn more about the increasing...
With 3D cell culture applications and bioprinting, cancer research is changing. Learn more about the increasing relevance of 3D bioprinting, how...
Using Organoids for Disease Modeling
For this Ask the Expert Session from The Cell Culture Dish, we have assembled a team of experts to answer your questions on disease modeling using organoids.
For this Ask the Expert...
For this Ask the Expert Session from The...
For this Ask the Expert Session from The Cell Culture Dish, we have...
For this Ask the Expert Session from The Cell Culture Dish, we have assembled a team of experts to answer your...
For this Ask the Expert Session from The Cell Culture Dish, we have assembled a team of experts to answer your questions on disease modeling...
Organoid Library Development: Testing Patient Sensitivity to Potential Cancer Therapies
Discover how one lab is using patient-derived pancreatic organoids to deliver personalized treatments.
Discover how one lab is using...
Discover how one lab is using...
Discover how one lab is using patient-derived pancreatic organoids to...
Discover how one lab is using patient-derived pancreatic organoids to deliver personalized treatments.
Discover how one lab is using patient-derived pancreatic organoids to deliver personalized treatments.

3D Bioprinting Revolutionizes Tissue Engineering
Take 3D cell culture research to a new level with Organovo’s 3D bioprinting technology and Corning’s Transwell permeable supports.
Take 3D cell culture research...
Take 3D cell culture research to a new level...
Take 3D cell culture research to a new level with Organovo’s 3D...
Take 3D cell culture research to a new level with Organovo’s 3D bioprinting technology and Corning’s Transwell...
Take 3D cell culture research to a new level with Organovo’s 3D bioprinting technology and Corning’s Transwell permeable supports.
Tissue Engineering: The Latest 3D Cellular Models for Respiratory Drug Development
Learn how a company in Switzerland is harnessing the latest cell culture technology to build even more predictive 3D cellular models for respiratory disease research.
Learn how a company in...
Learn how a company in Switzerland is...
Learn how a company in Switzerland is harnessing the latest cell culture...
Learn how a company in Switzerland is harnessing the latest cell culture technology to build even more predictive...
Learn how a company in Switzerland is harnessing the latest cell culture technology to build even more predictive 3D cellular models for...
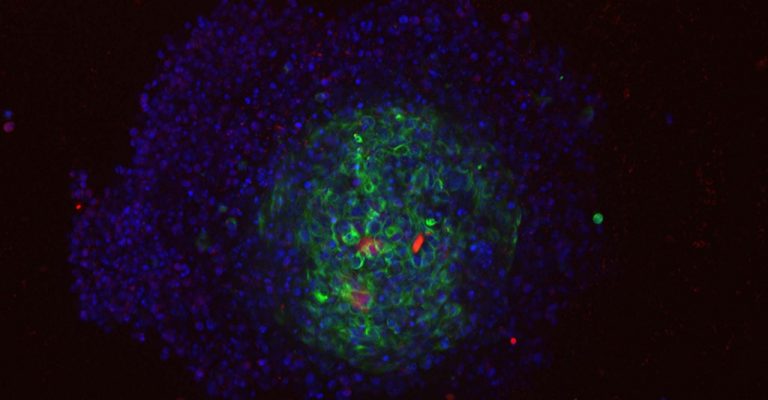
Advances in 3D Cell Culture for High Throughput Drug Screening
Dr. Timothy Spicer, Assistant Professor of Molecular Therapeutics and Lead ID Director of Discovery Biology & HTS at The Scripps Research Institute, Florida, USA talked to SelectScience about advances in cancer drug screening, being driven by 3D cell culture automation.
Dr. Timothy Spicer, Assistant...
Dr. Timothy Spicer, Assistant Professor of...
Dr. Timothy Spicer, Assistant Professor of Molecular Therapeutics and Lead...
Dr. Timothy Spicer, Assistant Professor of Molecular Therapeutics and Lead ID Director of Discovery Biology &...
Dr. Timothy Spicer, Assistant Professor of Molecular Therapeutics and Lead ID Director of Discovery Biology & HTS at The Scripps Research...
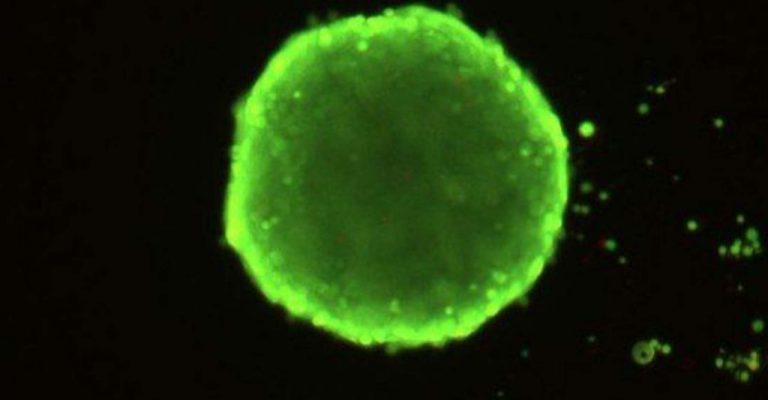
Ultra-high-throughput screening (uHTS): How scientists at Scripps Research target global medical breakthroughs
From assay inception to dose response and medicinal chemistry, discover the cutting-edge technology used to identify small molecules with clinical potential, in this SelectScience interview.
From assay inception to dose...
From assay inception to dose response and...
From assay inception to dose response and medicinal chemistry, discover...
From assay inception to dose response and medicinal chemistry, discover the cutting-edge technology used to...
From assay inception to dose response and medicinal chemistry, discover the cutting-edge technology used to identify small molecules with...
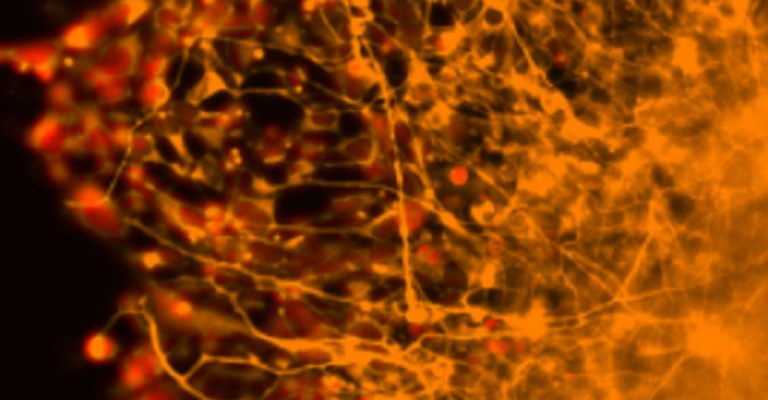
Scaling up with Spheroids Opens up New Possibilities in Cancer Therapy | 3D Cell Culture | Corning
Researchers at Uppsala University in Sweden develop innovative quiescent spheroid cell models and a unique process of high throughput screening to conduct drug-repositioning anti-cancer research.
Researchers at Uppsala...
Researchers at Uppsala University in Sweden...
Researchers at Uppsala University in Sweden develop innovative quiescent...
Researchers at Uppsala University in Sweden develop innovative quiescent spheroid cell models and a unique process...
Researchers at Uppsala University in Sweden develop innovative quiescent spheroid cell models and a unique process of high throughput screening...

Ottawa Researchers Conduct First Clinical Trial Using Stem Cell Therapy to Treat Septic Shock
Researchers from the Ottawa Hospital Research Institute successfully take their innovative research to the next level—scaling up to prepare for large-scale clinical research that may reframe the approach to treating patients with septic shock.
Researchers from the Ottawa...
Researchers from the Ottawa Hospital...
Researchers from the Ottawa Hospital Research Institute successfully take...
Researchers from the Ottawa Hospital Research Institute successfully take their innovative research to the next...
Researchers from the Ottawa Hospital Research Institute successfully take their innovative research to the next level—scaling up to prepare for...